Lisa Morere— MSN, APRN, ANP-BC—is the Clinical Lead at Outcomes4Me and is a board certified Adult Nurse Practitioner. She has over 21 years of experience in the healthcare setting, predominantly in oncology.
As a board certified Adult Nurse Practitioner, I have always valued the role that I have in people’s lives during the difficult journey of a cancer diagnosis. Throughout my clinical work, I’ve seen a lot of misconceptions floating around about how clinical trials work. It’s important to know that your participation in clinical trials will help you and help other people like yourself. It can be so rewarding to participate in a clinical trial after a cancer diagnosis. Not only are you getting a chance to experience new treatments, but you’re helping other cancer patients and are making an impact on cancer treatment as a whole.
I want to break down some common clinical trial myths I encounter and why these misconceptions are so common.
Myth #1 – Clinical Trial Volunteers/Participants are Human Guinea Pigs
The most common clinical trial myth I come across is that clinical trial volunteers or participants are treated like human guinea pigs. This myth is inaccurate because there is extensive oversight in clinical trials with both internal and government regulations to ensure research is guided by science and ethical principles. Patient safety is a top priority in clinical trials. The myth may stem from guinea pigs being commonly used as animal subjects for research in the past.
Today there are numerous guidelines in place surrounding clinical trial safety. For example Good Clinical Practice guidelines that follow a standard that protects the rights, well-being and safety of participants.
Myth #2 – Clinical Trials are Only Used as a Last Resort Option
Another common clinical trial myth I come across that is potentially dangerous, is that clinical trials are only used as a last resort treatment option. This belief may stem from clinical trials being an option when a patient has exhausted all other available treatments due to the treatment no longer working against the cancer, but this doesn’t mean that clinical trials can’t be used sooner in treatment.
Clinical trials can provide earlier access to new treatments. Clinical trials are available for different stages of cancer and a good time to look into clinical trial options is when treatment is working and cancer is responding to treatment as a way to be proactive. Additionally, some clinical trials are for supportive care to help ease the side effects of current treatments.
Myth #3 – Clinical Trials Only Use Placebos
Placebos can help researchers learn whether a new medicine works better than standard treatment and typically placebos are only used when no existing treatment for that cancer type exists. This doesn’t happen often at all due to standard of care being used as the control (IE: standard of care vs. standard of care plus placebo).
If a placebo is a possible treatment on a clinical trial, the participants will be informed of this prior to consenting or saying yes to participation.
Myth #4 – Clinical Trials Will Cause You to Lose Control of Your Personal Health Information
It’s also fairly common for people to falsely believe that clinical trials will cause you to lose control of your personal health information. It’s easy to see where this fear comes from, as the information on the outcomes of the clinical trial research is shared. Because of this, some worry that personal health information might be shared as well.
Fortunately, clinical trial participants don’t have to worry about their personal information being shared. An individual’s privacy is protected based on strict regulations—all of which are laid out in the informed consent process prior to participation.
Myth #5 – Once Enrolled in a Clinical Trial, I Can’t Change My Mind
Another major concern amongst cancer patients is that once they’re enrolled in a clinical trial, they won’t be able to change their mind and withdraw from the trial. The reason this is a common myth, is because people think that they have no other options and that they wouldn’t be allowed to stop participation. It can feel like you have a lack of control over your treatment plan, but that isn’t the case.
Participation in a clinical trial is voluntary. This is discussed at the time of consent. It’s important to discuss the desire to stop a treatment with your medical team so that it can be done safely and that other treatment options—if available—can be started in a timely fashion so that there is only a small gap in treatment.
Myth #6 – Clinical trials are dangerous
Because the word “investigational” is a part of research, some people feel that clinical trials are dangerous. This term can be misleading and cause people to think that this means trial and error is occuring during a clinical trial, but safety is the backbone of clinical trials. It is assessed on a constant basis by clinical research teams and participants throughout treatment and even after treatment stops (whether voluntarily or because the trial completed).
Myth #7 – Clinical Trials Are Expensive and Not Covered By Insurance
Understandably, many worry about the costs associated with a clinical trial. It’s very common to believe that their insurance won’t cover the costs of a clinical trial and it will be too expensive, as clinical trials are not “approved” treatments. However, federal law requires most health insurance plans to cover routine patient care costs in clinical trials under certain conditions, such as eligibility for the trial and a trial that has been approved for use. If a portion of the clinical trial is not covered by health insurance plans, often the trial sponsor will cover the costs. This information is a part of the discussion a patient has with clinical trial hosts prior to their participation, so there won’t be any surprise bills at the end.
Myth #8 – Clinical Trials Must Be Recommended to You By Your Treatment Team
An extremely common clinical trial myth I come across is that clinical trials must be recommended to you by your treatment team. Although it is important that your treatment team helps you navigate clinical trial options, they may not have the ability to know if you will be able to participate in a clinical trial.
Clinical research teams have had challenges during the pandemic and a lot of turnover, operating with less staff and resources creating longer wait times to enroll patients on clinical trials.
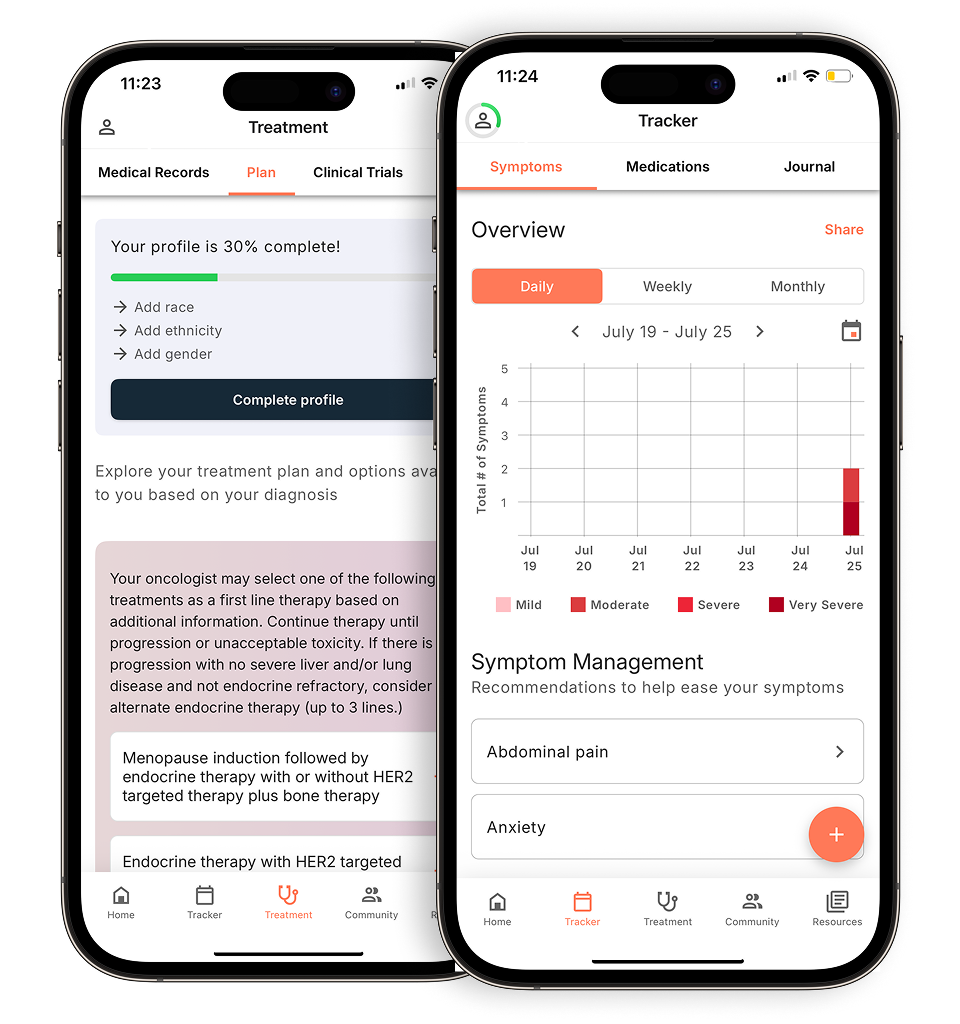
The best time to look into clinical trials is any time and you can do this on your own by using digital resources like Outcomes4Me’s Clinical Trial Finder to find clinical trials that may be a good fit for you.
Personalized support for real care decisions
Understand your diagnosis, explore clinical trials, and track symptoms--all in one place.
Get started
Compare treatments, prepare for appointments, and track side effects—all in the app
Built for your diagnosis, Outcomes4Me gives you the tools to make confident, informed decisions—right when you need them.
Continue in app