Outcomes4Me Secures $21M in Funding Learn more >>
Outcomes4Me Secures $21M in Funding Learn more >>
Suzanne Garner
Cancer Survivor, Outcomes4Me User

We’re the only DCIS HER2+ breast cancer app that integrates with the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®), from the not-for-profit alliance of 33 leading cancer centers. The NCCN Guidelines® include comprehensive guidance related to all FDA-approved treatment options.
We gather DCIS HER2+ breast cancer treatment recommendations typically meant for oncologists and use Artificial Intelligence (AI) to translate that information so you can understand it, putting you in control. With this knowledge, you can feel empowered to make the best medical decisions with your care team.
With our free DCIS HER2+ breast cancer app, you’ll get:
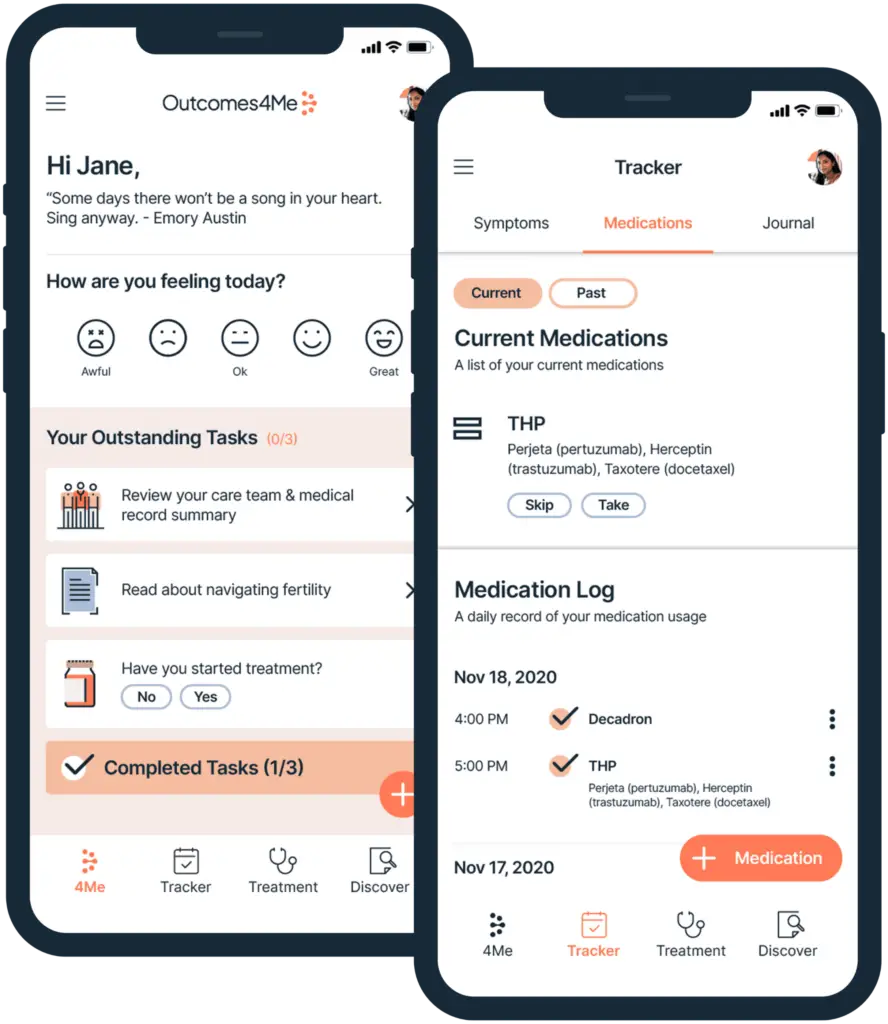

Gain direct access to personalized treatment options so you can have more informed conversations with your care team.

Rely on us to help your patients get access to the best DCIS HER2+ breast cancer care and resources, and save time in your day. We’re here to help and won’t ever get in the way.

Accelerate patient access to your treatments and expand the reach of your DCIS HER2+ breast cancer research and clinical trials.
Get all the information you need to take a more active role in treating your DCIS HER2+ breast cancer.












Yes, DCIS can be either Her2-negative or positive. However, at this time the prognostic significance of Her2 status in DCIS is not well established and as a result it is currently not recommended to test for Her2 status. HER2 testing is not used in the diagnosis and treatment of DCIS.
The prognostic significance of Her2 status in DCIS is not well established and targeted therapy is not recommended. Treatment for Her2 Positive DCIS therefore is based on hormone receptor status and patient factors and is not influenced by Her2 status.
DCIS is not tested for HER2 for two reasons: one, DCIS is more likely than invasive cancer to be HER2+ (we don’t know why); two, DCIS is not treated with HER2-targeted therapies, so testing DCIS for HER2 would not influence treatment choices.
Yes, most cases of DCIS are hormone receptor-positive. People with hormone receptor-positive DCIS may benefit from hormone therapy (tamoxifen or an aromatase inhibitor) [5,9,14-18].
Source: NCCN
https://www.nccn.org/patients/guidelines/content/PDF/stage_0_breast-patient.pdf
Source: NCCN Footnote (taken from provider guidelines)
The NCCN Panel recommends testing for ER status in order to determine the benefit of adjuvant endocrine therapy or risk reduction. This is in accordance with the American Society for Clinical Oncology (ASCO)/CAP guidelines,4 which recommend that ER testing of newly diagnosed DCIS to determine potential benefit of endocrine therapies for breast cancer risk reduction and progesterone receptor (PR) testing be considered optional. (NCCN Guideline quote)
Source: Dr Susan Love Foundation for Breast Cancer Research
https://drsusanloveresearch.org/ductal-carcinoma-situ-dcis/#:~:text=DCIS%20is%20not%20tested%20for,would%20not%20influence%20treatment%20choices.
Source: Susan Komen Foundation
https://www.komen.org/breast-cancer/treatment/by-diagnosis/dcis/#:~:text=Most%20cases%20of%20DCIS%20are,9%2C14%2D18%5D.
By clicking the “Subscribe Now” button you’re agreeing to our Terms of Service and Privacy Policy.