This week, the International Association for the Study of Lung Cancer (IASLC) hosted its annual World Conference on Lung Cancer in Singapore. Every year the conference brings together researchers, healthcare professionals, patients, and advocates to discuss the cutting-edge research happening in lung cancer diagnostics and treatment.
Staying apprised of the latest innovation in cancer research can be overwhelming, but patients deserve access to this information in order to make informed decisions about your care. Outcomes4Me recently expanded our platform to bring resources to non-small cell lung cancer (NSCLC) patients. As part of our efforts to keep you informed, we’ve rounded up four key research updates from this week’s World Conference on Lung Cancer.
1. Osimertinib Plus Chemo Now First-Line Standard in EGFR-Mutated NSCLC?
Data from the FLAURA2 clinical trial showed a significant increase in progression-free survival for patients with advanced EGFR-mutated NSCLC treated with osimertinib plus chemotherapy. Patients who were treated with osimertinib and chemotherapy had a 38% reduced risk of death or disease progression compared to those treated with osimertinib alone. Experts say these findings support the use of osimertinib plus chemo as a new first-line treatment option for patients with advanced EGFR-mutated NSCLC. Currently, EGFR tyrosine kinase inhibitors (TKIs) are the standard of care first-line treatment for this type of NSCLC.
2. Patritumab Deruxtecan May Benefit Patients With NSCLC and Few Options
Researchers from the Memorial Sloan Kettering Cancer Center presented findings from phase II of the HERTHENA-Lung01 trial that evaluated the efficacy of the antibody-drug conjugate (ADC) patritumab deruxtecan (HER3-DXd) in patients with advanced EGFR-mutated NSCLC. While researchers need more data to understand the overall survival benefit of HER3-DXd, the phase II results demonstrated clinically meaningful efficacy of the treatment, with an objective response rate (ORR) of 29.8% and median progression-free survival (PFS) of 5.5 months.
3. Iruplinalkib Produces Longer PFS Than Crizotinib in Advanced ALK+ NSCLC
According to data from the phase III INSPIRE trial, patients with ALK-positive advanced NSCLC had improved PFS and a higher ORR on iruplinalkib versus crizotinib. Patients on iruplinalkib experienced a median PFS of 27.7 months and an ORR of 93% as compared to 14.63 months and 89.3% respectively for patients being treated with crizotinib. Researchers from this study are hopeful that iruplinalkib will be a new treatment for patients with this subtype of NSCLC.
4. 2-Year Follow-Up Supports Adagrasib Monotherapy in KRAS G12C–Mutant NSCLC
Findings from a two-year follow-up of the phase 1 KRYSTAL-1 trial showed promising survival outcomes for patients with KRAS G12C–mutated advanced NSCLC being treated with adagrasib monotherapy. Based on the follow-up data, patients on adagrasib experienced a median overall survival of 14.1 months, with a 1-year PFS rate of 35% and 2-year PFS rate of 13.9%. During this phase 1 trial, the drug was well-tolerated and showed no new safety signals, so researchers will continue to evaluate efficacy in phase II.
Please reach out to your own provider with any questions. We also have a team of oncology nurse practitioners available to connect with you. Reach out via Ask Outcomes4Me.
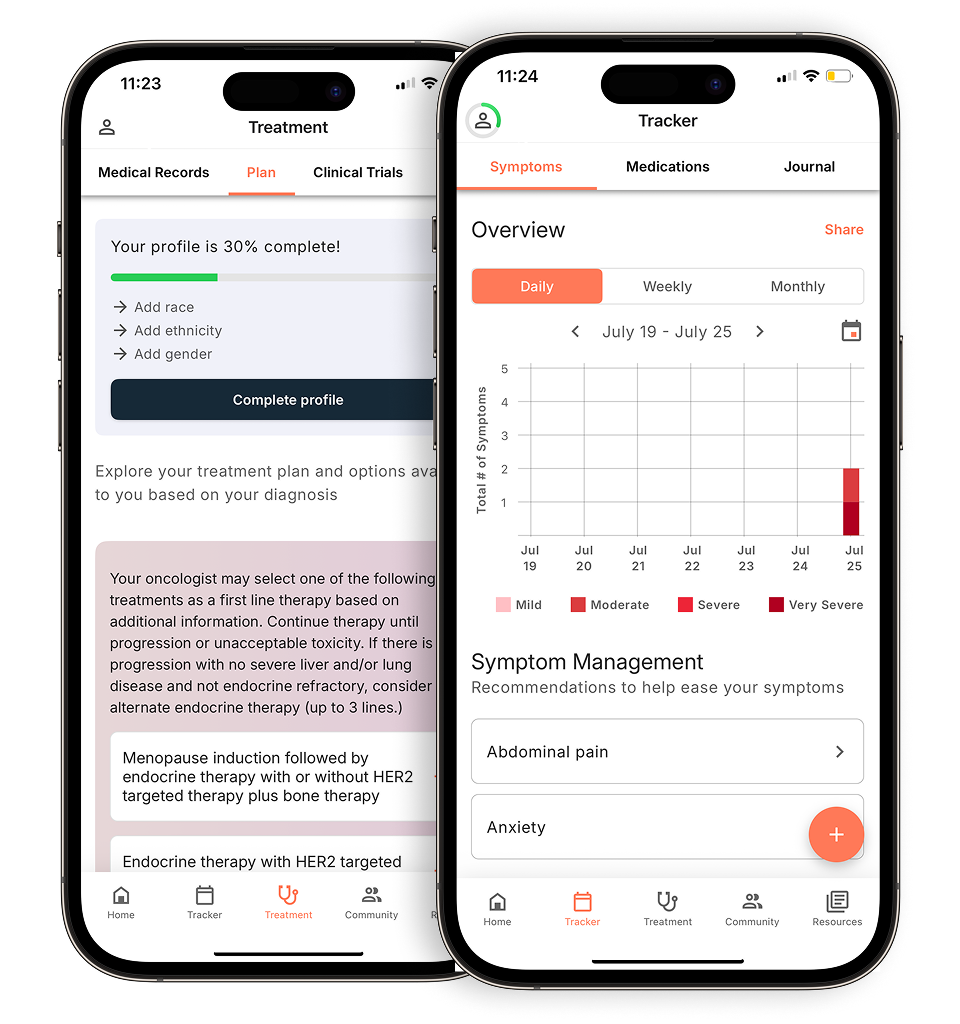
Personalized support for real care decisions
Understand your diagnosis, explore clinical trials, and track symptoms--all in one place.
Get started
Compare treatments, prepare for appointments, and track side effects—all in the app
Built for your diagnosis, Outcomes4Me gives you the tools to make confident, informed decisions—right when you need them.
Continue in app